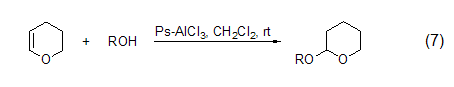3,4-Dihydro-2H-pyran 3,4-Dihydro-2H-pyran
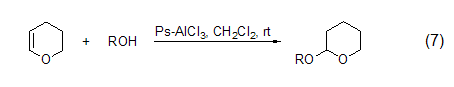
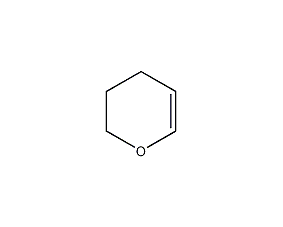
Structural formula
| Business number | 032A |
|---|---|
| Molecular formula | C5H8O |
| Molecular weight | 84.12 |
| label |
3,4-Dihydropyran, 2,3-Dihydropyran, 2,3-Dihydro-4H-pyran, 3,4-Dihydro-2H-pyran, Diihydropyran, Ether and acetal solvents, Heterocyclic compounds |
Numbering system
CAS number:110-87-2
MDL number:MFCD00006558
EINECS number:203-810-4
RTECS number:None
BRN number:103493
PubChem number:24863242
Physical property data
1. Properties: colorless, easy-flowing liquid
2. Relative density (g/mL, 20/4℃): 0.922
3. Relative vapor density (g/ mL, air = 1): 2.9
4. Melting point (ºC): -70
5. Boiling point (ºC, normal pressure): 86.5
6 . Refractive index (n20ºC): 1.4423
7. Flash point (ºC, closed): -15
8. Autoignition point or ignition temperature (ºC): 240
9. Saturated vapor pressure (kPa, 20ºC): 120
10. Explosion upper limit (%, V/V): 13.8
11. Explosion lower limit (%, V/V): 1.1
12. Solubility: Miscible with ethanol, ether and general organic solvents, slightly soluble in water.
13. Refractive index at room temperature (n25): 1.4400
14. Critical temperature (ºC): 288.85
15. Critical pressure (MPa): 4.56
16. Critical density (g·cm-3): 0.314
17. Critical volume (cm 3·mol-1): 268
18. Critical compression factor: 0.262
19. Eccentricity factor: 0.247
20. Freezing point (ºC): -15
21. Solubility: soluble in water, ethanol, slightly soluble in chloroform.
Toxicological data
1. Acute toxicity:
Oral LD50 in rats: >4000mg/kg;
Inhalation LC50 in rats: >10700mg/m3/4H
2. Irritation: Transdermal to rabbits, moderately irritating. Rabbit eye, moderate irritation.
Ecological data
This substance may be harmful to the environment, and special attention should be paid to the pollution of water bodies.
Molecular structure data
1. Molar refractive index: 17.20
2. Molar volume (cm3/mol): 70.6
3. Isotonic specific volume (90.2K ): 157.2
4. Surface tension (dyne/cm): 24.5
5. Polarizability (10-24cm3): 6.82
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 1
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: none
6. Topological molecule polar surface area 9.2
7. Number of heavy atoms: 6
8. Surface charge: 0
9. Complexity: 57
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocentersAmount: 0
12. Uncertain number of stereocenters of atoms: 0
13. Determined number of stereocenters of chemical bonds: 0
14. Uncertain chemical bonds Number of stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. It does not decompose under normal temperature and pressure. Avoid contact with strong oxidants and heat. It is flammable and can cause combustion or explosion when exposed to high heat, open flames and oxidants.
2. When in contact with peroxide, sensitive peroxide is generated. Purification of these compounds can cause violent explosions and great care must be taken in isolating these products.
3. Existence form: Found in flue-cured tobacco leaves.
Storage method
Store in a cool, ventilated warehouse. Keep away from fire, heat sources and anti-static. The storage temperature should not exceed 30℃. should be kept away from oxidizer, do not store together. Use explosion-proof lighting and ventilation facilities. It is prohibited to use mechanical equipment and tools that are prone to sparks. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method
It is obtained by dehydration and ring expansion of tetrahydrofurfuryl alcohol.
Purpose
1. Tetrahydropyran, pentanediol, glutaric acid, valerolactone, pentadiene and resin products can be produced through polymerization, hydrogenation, oxidation and other reactions. It can also be used as pharmaceutical intermediates. Solvents, organic synthesis intermediates.
2. It is widely used for the protection of hydroxyl groups, generally does not react with nucleophiles and organometallic reagents, is resistant to strong alkali, and is easy to change under low pH or Lewis acid conditions.
Tetrahydropyranation of alcohols The protection of alcohols by tetrahydropyran (THP) is a very useful tool in organic synthesis. The reaction of chiral alcohols with dihydropyrans introduces an additional asymmetric center, thus yielding a diastereomeric mixture (Formula 1). This brings certain difficulties to purification and spectral analysis, but it does not prevent its successful application[1].
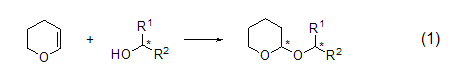
Under very mild reaction conditions (0 oC, 1 h, 89%~100%), (trimethylsilyl) sulfate can be used to cause tetrahydropyranylation of alcohols, that is, allyl tertiary alcohols are used There will be no rearrangement reactions.
Decomposition of tetrahydropyran derivatives In the presence of MEM ether, MOM ether and 1,3-dioxolane, the organotin phosphate condensation reaction is important for TEM The selective splitting of ether is very efficient and the catalyst can be reused. In tert-butyldisilyl, acetyl, methanesulfonyl and methoxymethyl ether and in methanesulfonyl catalyst Me2Sn(SMe)2-BF3·Et2O, THP ethers of primary, secondary and tertiary alcohols will undergo selective cleavage [2].
Tetrahydropyranation of thiols Tetrahydropyran derivatives of thiols can be used in the shielding reaction of functional groups (Formula 2).
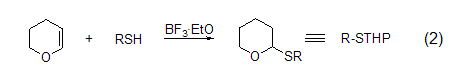
with O-tetrahydropyran ether, S-tetrahydropyran ether can also exist stably under the condition of 4 mol/L HCl-MeOH. Deprotection reactions using silver nitrate or hydrogen bromide-trifluoroacetic acid are easy to perform and give high yields. Oxidation reactions can occur with iodine, cyanogen sulfide, and disulfide[3].
Tetrahydropyranation of amines In the presence of a catalytic amount of p-toluenesulfonic acid, purine reacts with dihydropyran to form 9-(2-tetrahydropyran) Derivatives (Formula 3)[4].
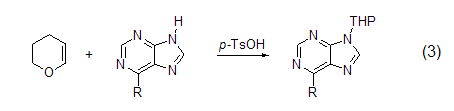
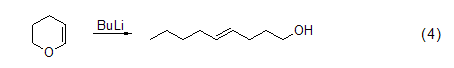
Dihydropyran The ring-opening reaction of dihydropyran treated with sodium n-pentyl or n-butyllithium produces trans-4-hydroxy-1-ol (formula 4)[5].
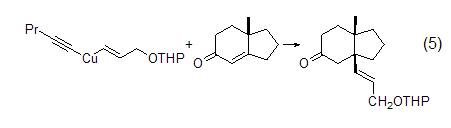
In organic synthesis, THP is used Protected groups have a wide range of applications. The structure of cis-CH=CHCH2OH (Formula 5)[6] can be introduced using Gilman cuprate reagent with chain-end protecting ether.
Dihydropyran and BrCH2C(NOH)CO2Et can be used in the synthesis of aminotriesters (Formula 6) [7].
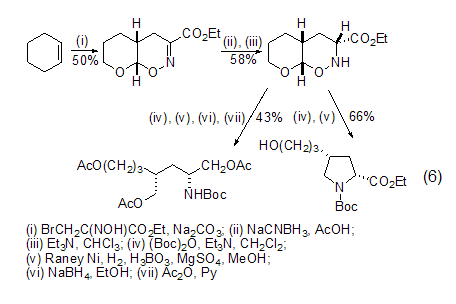
A simple and effective high chemistry option The tetrahydropyranylation method of alcohols and phenolic compounds is carried out at room temperature under the action of polystyrene with a catalytic amount of AlCl3 as a carrier. This method is highly selective for the monoprotection of symmetric diols (Formula 7)[8].