5-Ethyl-5-phenyl-dihydro-4,6(1H,5H)-pyrimidinedione 5-Ethyldiodro-5-phenyl-4,6(1H,5H)-pyrimidinedione
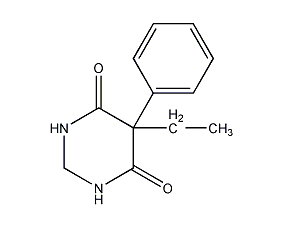

Structural formula
| Business number | 03JR |
|---|---|
| Molecular formula | C12H14N2O2 |
| Molecular weight | 218.26 |
| label |
2-Desoxyphenobarbital, 5-Ethyl-5-phenylhexahydropyrimidine-4,6-dione, aromatic compounds |
Numbering system
CAS number:125-33-7
MDL number:MFCD00038662
EINECS number:204-737-0
RTECS number:UV9100000
BRN number:None
PubChem number:24277732
Physical property data
1. Properties: white crystalline powder. Odorless, slightly bitter taste.
2. Melting point: 281-282℃
3. Solubility: slightly soluble in alcohol, almost insoluble in water, acetone or benzene
4. Water-soluble Properties: <0.1 g/100 mL (19℃)
Toxicological data
None
Ecological data
None
Molecular structure data
1. Molar refractive index: 59.08
2. Molar volume (cm3/mol): 191.7
3. Isotonic specific volume (90.2K): 477.6
4. Surface tension (dyne/cm): 38.5
5. Polarizability (10-24cm3): 23.42
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 2
3. Number of hydrogen bond acceptors: 2
4. Number of rotatable chemical bonds: 2
5. Number of tautomers: 3
6. Topological molecule polar surface area 58.2
7. Number of heavy atoms: 16
8. Surface charge: 0
9. Complexity: 279
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
Basic properties: The anti-epileptic effect is similar to that of phenobarbital, but more selective than phenobarbital; the intensity of effect is not as strong as that of phenytoin, but the combination of the two has a synergistic effect.
Storage method
Storage: sealed and stored.
Synthesis method
Brief description of the production method: Add ethyl acetate to sodium ethoxide, reflux for half an hour, cool to room temperature, add diethyl ethylphenylmalonate and thiourea, and flow for 9 hours. Cool to 0℃, add ethanol, and drop hydrochloric acid at 0-5℃ until pH=3-3.5. Add zinc powder, raise the temperature to reflux, slowly add hydrochloric acid to keep the solution with 2-3% acid content, complete the addition, and keep warm for 3 hours. Filter while hot, and recover ethanol from the filtrate. coldto 20°C, add water, filter, and wash the filter cake to obtain the finished product.
Purpose
Usage: Used for grand mal epilepsy and psychomotor seizures.
