Trimethylborate Trimethylborate
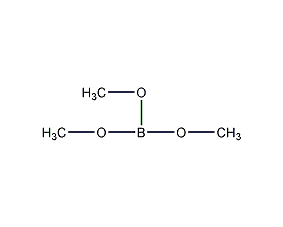
Structural formula
| Business number | 03DJ |
|---|---|
| Molecular formula | C3H9BO3 |
| Molecular weight | 103.91 |
| label |
(Tri)methyl borate, trimethoxyborane, Trimethyl orthoborate, Trimethylborate (methyl borate), Methyl borate, Boron trimethoxide, Boric acid trimethyl ester, Boron methoxide, Trimethoxyborane, Vulcanizing agent, wood preservatives, catalyst, gelling agent, Heat stabilizers |
Numbering system
CAS number:121-43-7
MDL number:MFCD00008346
EINECS number:204-468-9
RTECS number:ED5600000
BRN number:1697939
PubChem ID:None
Physical property data
1. Properties: colorless liquid.
2. Relative density (20/4ºC): 0.915
3. Refractive index (25ºC): 1.3548
4. Flash point (ºC): 29
5. Melting point (ºC): -34
6. Boiling point (ºC): 68-69
7. Solubility: soluble in tetrahydrofuran, ether , methanol and other organic solvents.
Toxicological data
1. Acute toxicity: Rat passing LD50: 6140mg/Kg; Rats passing the peritoneal cavity LDL0: 1600mg/Kg
mice LD50: 1290 mg/kg; Cavity LDL0: 1gm/kg
Rabbit skin LD50: 1980uL/kg; rabbit transperitoneal LDL0: 1600 mg/kg
2. Irritating to the eyes and respiratory system, and not related to the skin Harmful by contact.
Ecological data
None
Molecular structure data
1. Molar refractive index: 24.61
2. Molar volume (cm3/mol): 119.0
3. Isotonic specific volume (90.2K): 247.4
4. Surface tension (dyne/cm): 18.6
5. Dielectric constant:
6. Dipole moment (10-24 cm3):
7. Polarizability: 9.75
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 3
4. Number of rotatable chemical bonds: 3
5. Number of tautomers: none
6. Topological molecule polar surface area 27.7
7. Number of heavy atoms: 7
8. Surface charge: 0
9. Complexity: 31.7
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Moisture-sensitive liquid smokes in the air. Very flammable, flash point -1℃. Miscible with ether, methanol, tetrahydrofuran, isopropylamine and hexane. Stable in anhydrous state, it decomposes into methanol and boric acid when exposed to water.
Storage method
Store sealed in a cool, dry place.
Synthesis method
1. It can be synthesized by various methods, such as from pyridine-boron trifluoride complex, or from methanol; boron fluoride; methanol; boric acid; methyl orthosilicate; boron halide, etc.
2.The boron oxide obtained by melting boric acid in a platinum container is quickly cooled and pulverized, and then stored in a desiccator with phosphorus pentoxide.
Instruments; mounting as shown. The bottleneck of the three-neck round bottom flask has a ground mouth and a sliding head. The rubber plug with glass container R (for holding boron oxide) is in A. The lower end of the container is opened and ground so that it can be closely connected upward with the leading glass ball valve. The cross-wire stirrer E is used to break up lumps that may form near the outlet of the container. The B neck of the flask is connected to the condenser, and the upper end is welded to the U-shaped tube used to hold calcium chloride. A rubber stopper with a curved glass rod F inserted into neck C serves as a stirrer.
When preparing methyl borate, close the ball valve of R. Put 75 grams of finely powdered boron oxide in R. Put 320 ml of absolute methanol from the Bneck and connect it to the condenser. No. i-1, loosen the ball valve D slightly, and the boron oxide will fall into the methanol at any required speed. This will facilitate the control of the exothermic reaction process to avoid the reaction being too intense. After adding boron, rotate the glass rod F back and forth to stir the solution.
The boron oxide should be added quickly enough to keep the mixture hot, but not so fast that it causes the solution to boil. This feeding process takes about 10 minutes. The reaction mixture was allowed to cool, and snow-white crystalline boric acid precipitated. Pour off the turbid liquid and distill it with a Johnson fractionating column to obtain 175 ml of azeotropic mixture of methyl borate and methyl alcohol. Use cold concentrated sulfuric acid treatment solution to remove formazan-o and discard the bottom layer. The methyl borate is treated with sulfuric acid twice, fractionated with Johnson column, and vacuum fractionated again. Finally, the pure product is obtained with a yield of 42%
Use A similar method can be used to prepare ethyl borate with a yield of 31%
Install a fractionation column with a diameter of 2.5 meters and a length of 75 centimeters on a 1-liter round-bottomed flask, and fill it with a 4.} mm stainless steel spiral to achieve a theoretical efficiency of 15. Put 0.5 moles of borax (Na2B4O210H2O, 16 moles of methanol and 1 mole of sulfuric acid into the bottle. Slowly distill the reaction mixture to azeotrope J. at 54°C to obtain most of the product, and a small part can be obtained at 54–62°C. Obtained. Yield 92.9%
![]()
3. Preparation method:
Equipped with a stirrer and fractionating device (fractionating column has a diameter of 2.5cm, a height of 75cm, and a stainless steel spiral ring inside. , to achieve the effect of 15 theoretical plates), add 0.5 mol of borax, 16 mol of methanol, and 1 mL of sulfuric acid, slowly heat to reflux, and collect most of the constant boiling point sulfur products at 54°C, and a small amount of 54 Distillate at ~62°C, the total yield of trimethyl borate is 92.9%.[3]
Purpose
1. Intermediate of potassium (sodium) boron hydrogen. Vulcanizing agent, wood preservative, catalyst, gelling agent, heat stabilizer, hydrogen flame fire extinguishing agent, also used for flame retardant treatment of cotton and preparation of active silica, and as reagent for gas chromatography analysis of carbohydrate derivatives. Solvent for paraffins, resins and petroleum.
2.Aromatic halide reacts with methyl borate through Grignard reagent and is hydrolyzed to generate phenol. An accelerator for the diborane reaction.
