Fluorene
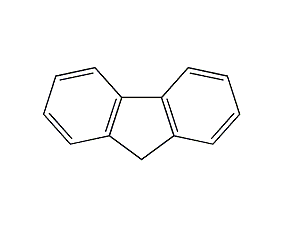

Structural formula
| Business number | 01X0 |
|---|---|
| Molecular formula | C13H10 |
| Molecular weight | 166 |
| label |
9H-fluorene, 2,3-Benzindene, o-Diphenylmethane, 9H-Fluorene, 2,3-Benzindene, o-Biphenylenemethane, aromatic compounds |
Numbering system
CAS number:86-73-7
MDL number:MFCD00001111
EINECS number:201-695-5
RTECS number:LL5670000
BRN number:1363491
PubChem number:24847846
Physical property data
1. Properties: white flake crystals, fluorescent when impure
2. Density (g/mL, 25/4℃): 1.203
3. Relative Density (20℃, 4℃): 1.208
4. Melting point (ºC): 114.77
5. Boiling point (ºC, normal pressure): 294℃
6. Solubility parameter (J·cm-3)0.5: 21.646
7. van der Waals area (cm2·mol-1): 1.019×1010
8. Flash point (ºC): 151℃
9. van der Waals volume (cm3·mol-1): 93.670
10. Autoignition point or ignition temperature (ºC): Uncertain
11. Vapor pressure (kPa, 25ºC): Uncertain
12. Saturated vapor pressure (kPa, 60ºC): Uncertain
13. Heat of combustion (KJ/mol): Uncertain
14. Critical temperature (ºC): Uncertain
15. Critical pressure (KPa): Uncertain
16. The logarithmic value of the oil-water (octanol/water) partition coefficient: Uncertain
17. The upper limit of explosion (%, V/V): Uncertain
18. The lower limit of explosion (%, V/V): Uncertain
19. Solubility: Insoluble in water, soluble in ethanol, ether, benzene, carbon disulfide and other organic solvents
Toxicological data
1. Acute toxicity:
Mouse abdominal cavity LD50: >2mg/kg;
2. Teratogenicity
Mouse lymphocytes: 19500 nmol/L; mouse mammary gland: 1 ug/L;
Mouse lymphocytes: 150umol/L; mouse lung: 25mg/L;
Ecological data
None yet
Molecular structure data
1. Molar refractive index: 53.79
2. Molar volume (cm3/mol): 148.3
3. Isotonic specific volume (90.2K): 386.7
4. Surface tension (dyne/cm): 46.2
5. Polarizability (10-24cm3): 21.32
Calculate chemical numbersAccording to
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 0
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: none
6. Topological molecule polar surface area 0
7. Number of heavy atoms: 13
8. Surface charge: 0
9. Complexity: 165
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. This product is toxic. Mice were intraperitoneally injected with LD50260mg/kg. Operators should wear protective equipment.
2. Exist in smoke.
3. Easy to sublimate in high vacuum.
Storage method
This product should be sealed and stored in a cool place. This product is packed in plastic bags.
Synthesis method
1. The fluorene content in high-temperature tar is about 1.0-2.0%. Cut the 290-310°C fluorene fraction from the wash oil fraction, use a distillation tower equivalent to 115 theoretical plates for precise fractionation, the reflux ratio is 15:1, and cut the 292-302°C narrow fraction. Through cooling, crystallization, and centrifugal separation, crude fluorene is obtained, and then recrystallized with 1:25 benzene or xylene to obtain industrial fluorene. Dissolve industrial fluorene in benzene, wash with 40% sulfuric acid, then wash with neutralizing water, remove the solution, re-distill the fluorene, and recrystallize the distillate with gasoline and ethanol to obtain pure fluorene with a purity of 95%.
2. It is produced by reacting biphenyl with methylene chloride under the catalytic action of ferric chloride.
Purpose
Used as raw material for organic synthesis. It can be made into trinitrofluorenone, which is used for electrostatic printing; it can be used to synthesize aryl transparent nylon; it can replace anthraquinone to synthesize indanthrene dye; it can be used to make antispasmodics, sedatives, analgesics, and antihypertensive drugs; it can be synthesized Pesticides, herbicides; preparation of impact-resistant organic glass and fluorenal resin; also used as wetting agents, detergents, liquid flash agents, disinfectants, etc.
