Ethyl Formate Ethyl Formate
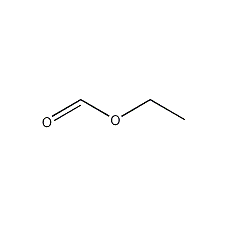

Structural formula
| Business number | 0306 |
|---|---|
| Molecular formula | C3H6O2 |
| Molecular weight | 74.08 |
| label |
Ethyl formate, Ethyl formate, Formic acid ethyl ester, Formic ether, Ethyl methanoate, fungicides, larvicides, fumigant, food additives, Flavor enhancer |
Numbering system
CAS number:109-94-4
MDL number:MFCD00003294
EINECS number:203-721-0
RTECS number:LQ8400000
BRN number:906769
PubChem number:24889112
Physical property data
1. Properties: colorless, easy-flowing liquid with aromatic odor. [1]
2. Melting point (℃): -80[2]
3. Boiling point (℃): 54.3[3]
4. Relative density (water = 1): 0.92[4]
5. Relative vapor Density (air=1): 2.55[5]
6. Saturated vapor pressure (kPa): 25.6 (20℃)[6]
7. Heat of combustion (kJ/mol): -1630[7]
8. Critical temperature (℃): 235.3[8]
9. Critical pressure (MPa): 4.74[9]
10. Octanol/water partition coefficient: 0.23 [10]
11. Flash point (℃): -20 (CC) [11]
12. Ignition temperature ( ℃): 455[12]
13. Explosion upper limit (%): 16.5[13]
14. Explosion Lower limit (%): 2.7[14]
15. Solubility: Slightly soluble in water, soluble in most organic solvents such as benzene, ethanol, and ether. [15]
16. Viscosity (mPa·s, 15ºC): 0.419
17. Heat of evaporation (KJ/mol, 54.15ºC): 30.15
18. Heat of formation (KJ/mol): 369.28
19. Specific heat capacity (KJ/(kg·K), 15~30ºC, constant pressure): 2.00
20. Boiling point rising constant: 2.086
21. Conductivity (S/m, 20ºC): 1.45×10-9
22. Thermal conductivity (W/(m·K), 12ºC): 0.1583
23. Volume expansion coefficient (K-1, 20ºC): 0.00141
24. Critical density (g·cm-3): 0.323
25. Critical volume (cm3·mol-1): 229
26. Critical compression factor: 0.257
27. Eccentricity factor: 0.285
28. Lennard-Jones parameters (A): 11.14
29. Lennard-Jones parameter (K): 161.3
30. Solubility parameter (J·cm-3)0.5: 18.939
31. van der Waals area (cm2·mol-1): 6.440×109 sup>
32. van der Waals volume (cm3·mol-1): 42.740
33. Gas phase standard Heat of combustion (enthalpy) (kJ·mol-1): -1639.7
34. Gas phase standard claims heat (enthalpy) (kJ·mol-1): -398.3
35. Liquid phase standard combustion heat (enthalpy) (kJ·mol-1): -1607.5
36. Liquid phase Standard claims heat (enthalpy) (kJ·mol<After esterification by heating under ambient conditions, it is obtained by dehydration and distillation with phosphorus pentoxide or anhydrous calcium chloride.
Refining method: Since ethyl formate is easily hydrolyzed, it often contains free formic acid and ethanol and is acidic. During refining, it is first washed with sodium carbonate solution, then dried and fractionated with anhydrous potassium carbonate or phosphorus pentoxide. Calcium chloride forms a crystalline complex with ethyl formate and is not suitable for use as a desiccant.
4. Preparation method:
In a reaction bottle equipped with a reflux condenser, add 61g of 98% formic acid (1.33mol) and 31g of absolute ethanol (2) (0.67 mol), one particle of zeolite, heated to reflux for 24 hours. Change to a distillation device for distillation, collect fractions below 62°C, saturate the leachate with sodium chloride, and then wash with saturated sodium bicarbonate and saturated sodium chloride solutions in sequence. Dry with anhydrous sodium sulfate, fractionate, and collect the fraction at 53-54°C to obtain 36 g of butyl formate (1), with a yield of 72%. [25]
Purpose
1. Ethyl formate is used as a solvent for nitrocellulose, cellulose acetate, etc., as well as a fungicide, larvicidal and fumigant for food, cigarettes, cereals, dried fruits, etc. As a flavor, ethyl formate can be used to blend the aromas of peaches, bananas, apples, apricots, pineapples, berries, etc. It can also be used as a spice for butter, brandy, liqueur, whiskey, etc. Ethyl formate is also an intermediate in organic synthesis. For example, in the pharmaceutical industry, it is used in the production of anti-tumor drugs Fulcedine and vitamin B1; ethyl formate and acetone are condensed in sodium methoxide-xylene solution to obtain sodium acetyl vinyl alcohol, which is condensed with acetyl glycine ester and then reacted with thiocyanate Cyclization with potassium can give 2-mercaptoimidazole-4-carboxylic acid ethyl ester. Ethyl formate is used to produce drugs such as Houttuynia cordata, Tongjingning, Anadrol, pyrantel, pyridine, Lixuesheng, and fully synthetic anisodamine.
2. Used as a solvent for cellulose acetate and cellulose nitrate, as well as for flavor synthesis and pharmaceutical production. [24]
