3,5-Dinitrobenzamide 3,5-Dinitrobenzamide
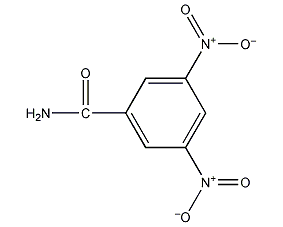

Structural formula
| Business number | 03E3 |
|---|---|
| Molecular formula | C7H5N3O5 |
| Molecular weight | 211.15 |
| label |
dinitrobenamide, nitromete, 3,5-Dinitrobenzamide, nitramine, Nitromid, Nitromide, 3,5-dinitro-benzamid, Benzamide, 3,5-dinitro-, component of Tristat, component of Unistat, component of Unistat-3, Tristat, Fragrance |
Numbering system
CAS number:121-81-3
MDL number:MFCD00007985
EINECS number:204-499-8
RTECS number:CV4752300
BRN number:7096825
PubChem number:24893576
Physical property data
1. Melting point (℃):183℃
2. Solubility:Slightly soluble in cold water, slightly more soluble in hot water.
Toxicological data
1, acute toxicity: rat oral LD50: 1250 mg/kg
Rat transperitoneal cavity LD50: 320mg/kg
Chicken orally LD50: 900mg/kg
Chicken transperitoneal cavity LD50: 280mg/kg
Turkey Oral LD50: 1150mg/kg
Ecological data
None
Molecular structure data
5. Molecular property data: 1. Molar refractive index:48.27 2. Molar Volume(m3/mol):131.8 3. Isotonic specific volume(90.2K):392.6 4. Surface tension(dyne/cm):78.7 5. Dielectric constant: 6. Dipole moment(10(m3/mol):131.8 3. Isotonic specific volume(90.2K):392.6 4. Surface tension(dyne/cm):78.7 5. Dielectric constant: 6. Dipole moment(10-24cm3) : 7. Polarizability:19.13
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 5
4. Number of rotatable chemical bonds: 1
5. Number of tautomers: 2
6. Topological molecule polar surface area 135
7. Number of heavy atoms: 15
8. Surface charge: 0
9. Complexity: 272
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
None
Storage method
None
Synthesis method
1. Introduction to production methods
Use3,5-Prepared from the reaction of dinitrobenzoyl chloride and ammonium acetate.
Purpose
2. Purpose
Used as anticoccidial drug. The advantage is that resistance develops slowly
= ST1 />– 24cm3):
7. Polarizability:19.13
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 5
4. Number of rotatable chemical bonds: 1
5. Number of tautomers: 2
6. Topological molecule polar surface area 135
7. Number of heavy atoms: 15
8. Surface charge: 0
9. Complexity: 272
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
None
Storage method
None
Synthesis method
1. Introduction to production methods
Use3,5-Prepared from the reaction of dinitrobenzoyl chloride and ammonium acetate.
Purpose
2. Purpose
Used as anticoccidial drug. The advantage is that resistance develops slowly