Levulinic acid Levulinic acid
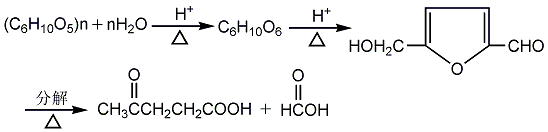
Structural formula
| Business number | 03H3 |
|---|---|
| Molecular formula | C5H8O3 |
| Molecular weight | 116.12 |
| label |
4-keto-n-pentanoic acid, 4-oxo-pentanoic acid, γ-oxopentanoic acid, 4-Oxopentanoic acid, 4-Oxovaleric acid, γ-Ketovaleric acid, Laevulinic acid, Multifunctional solvents, extraction separation agent, acidic solvent |
Numbering system
CAS number:123-76-2
MDL number:MFCD00002796
EINECS number:204-649-2
RTECS number:OI1575000
BRN number:506796
PubChem number:24882128
Physical property data
1. Properties: White flake or leaf-like crystals
2. Relative density (g/mL, 20/4ºC): 1.1335
3. Relative density (g /mL, 25/4ºC): 1.1447
4. Refractive index (25ºC): 1.4796
5. Refractive index (20ºC): 1.4396
6. Boiling point (ºC, 101.3kPa): 245~246 (dec)
7. Flash point (ºC): 138
8. Melting point (ºC): 33~35
9. Heat of evaporation (KJ/mol, 149.5ºC): 68.02
10. Heat of fusion (KJ/mol): 9.23
11. Solubility: easily soluble in water, Organic solvents such as alcohols and ethers. Insoluble in non-polar organic solvents such as gasoline, kerosene, and carbon tetrachloride.
Toxicological data
1. Skin/eye irritation: Human skin standard Drez eye dye test: 500mg/24H has a moderate irritating effect on the skin.
2. Acute toxicity: rat oral LD50: 1850mg/kg; mouse peritoneal LD5O: 450mg/kg
Rabbit skin LD50: >5gm/kg
Ecological data
Slightly harmful to water bodies.
Molecular structure data
1. Molar refractive index: 26.80
2. Molar volume (cm3/mol): 102.8
3. Isotonic specific volume (90.2K ): 257.9
4. Surface tension (dyne/cm): 39.6
5. Polarizability (10-24cm3): 10.62
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 3
p>
4. Number of rotatable chemical bonds: 3
5. Number of tautomers: 3
6. Topological molecule polar surface area 54.4
7. Number of heavy atoms: 8
8. Surface charge: 0
9. Complexity: 106
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. White flake or leaf-like crystals, flammable. Easily soluble in water; alcohol; ether organic solvents; insoluble in aliphatic hydrocarbons. It hardly decomposes under normal pressure distillation. If heated for a long time, it will lose water and generate unsaturated γ-lactone. It is hygroscopic and its aqueous solution is more acidic than acetic acid.
2. Chemical properties: Ketone reactions and acid reactions can occur. It reacts with hydrazine to form hydrazone; reacts with alkali to form salt; reacts with alcohol to form ester, etc. Oxidation, reduction, and halogenation reactions can also occur.
3. Exist in flue-cured tobacco leaves, burley tobacco leaves and smoke.
Storage method
Packed in plastic barrels and reinforced with wooden boxes. Store in a cool, dry and ventilated place. Keep away from fire and heat sources.
Synthesis method
1. Levulinic acid can be obtained by hydrolyzing the residue (furfural residue) from cottonseed shells or corncobs after producing furfural or waste potato residue with dilute acid under pressure. Add the uronic residue to 10% dilute hydrochloric acid, and the solidification liquid is 1:1.75. Mix evenly and then put it into the hydrolysis pot and cook it with steam at a pressure of 0.2MPa for 8-10 hours. Then filter the hydrolyzed dilute solution and concentrate it to a concentration of about 50%. Then carry out vacuum distillation and collect the fractions above 130℃ (2.67kPa) to obtain the finished product. Raw material consumption quota: potato poundage 7000kg/t; hydrochloric acid (100% discount) 1800kg/t. Another preparation method is obtained by rearrangement and hydrolysis of sugar alcohols. Add 4% hydrochloric acid solution to the reaction pot, stir and heat to 97-100°C, and slowly add the mixture of furfuryl alcohol; ethanol; and water. After adding, stir for another 30 minutes. Filter, and the filtrate is concentrated under reduced pressure at 80°C (21.3kPa) to obtain crude levulinic acid liquid. Then distill under reduced pressure, collect the 160-170°C (2.67kPa) fraction, and distill it again to obtain refined levulinic acid, with a yield of about 75%.
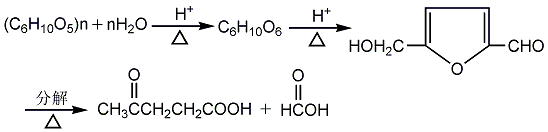
2. Tobacco: FC, 50 ;BU, 26.
Purpose
This product is mainly used as a raw material for manufacturing resins, medicines, spices and coatings. In the pharmaceutical industry, its calcium salt can be made into intravenous injections and indomethacin. Its lower esters can be used as food flavors and tobacco flavors. The bisphenolic acid made from this product can produce water-soluble resin, which is used in the paper industry to produce filter paper. It can also be used to make pesticides, dyes and surfactants. Also used as extraction and separation agent for aromatic compounds.