lidocaine
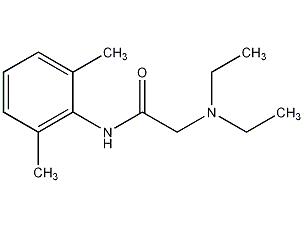

Structural formula
| Business number | 03RP |
|---|---|
| Molecular formula | C14H22N2O |
| Molecular weight | 234.34 |
| label |
N-diethylaminoacetyl-2,6-dimethylaniline, xylocaine, Xylocaine, lidocaine base, N-Diethylacetyl-2,6-dimethylaniline, 2-diethylamino-N-(2,6-dimethylphenyl)acetamide, 2-Diethylamino-N-(2,6-dimethylphenyl)acetamide, Lignocaine, Xylocaine, aromatic compounds |
Numbering system
CAS number:137-58-6
MDL number:MFCD00026733
EINECS number:205-302-8
RTECS number:AN7525000
BRN number:None
PubChem number:24896480
Physical property data
1. Appearance: white crystalline powder
2. Melting point (℃): 76~79
3. Solubility: easily soluble in water or ethanol, soluble in chloroform , insoluble in ether.
Toxicological data
None
Ecological data
None
Molecular structure data
1. Molar refractive index: 72.42
2. Molar volume (cm3/mol): 228.3
3. Isotonic specific volume (90.2K): 571.1
4. Surface tension (3.0 dyne/cm): 39.1
5. Polarizability (0.5 10-24cm3): 28.71
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 2
4. Number of rotatable chemical bonds: 5
5. Number of tautomers: 3
6. Topological molecule polar surface area 32.3
7. Number of heavy atoms: 17
8. Surface charge: 0
9. Complexity: 228
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
None
Storage method
This product should be sealed and stored in a cool, dry place.
Synthesis method
1. Chloroacetyl chloride method
2,6-Dimethylaniline undergoes chloroacetylation with chloroacetyl chloride, then condenses with diethylamine, and then acidifies to form a salt to obtain lidocaine.
2. Diethylaminoacetic acid methyl ester method
In the presence of sodium methoxide, 2,6-dimethylaniline and N,N-diethylaminoacetic acid methyl ester are condensed, It is then acidified to form a salt to obtain the product.
Purpose
Widely used for topical anesthesia, infiltration anesthesia, conduction anesthesia, and epidural anesthesia. It is also an important drug in the treatment of sudden arrhythmias.